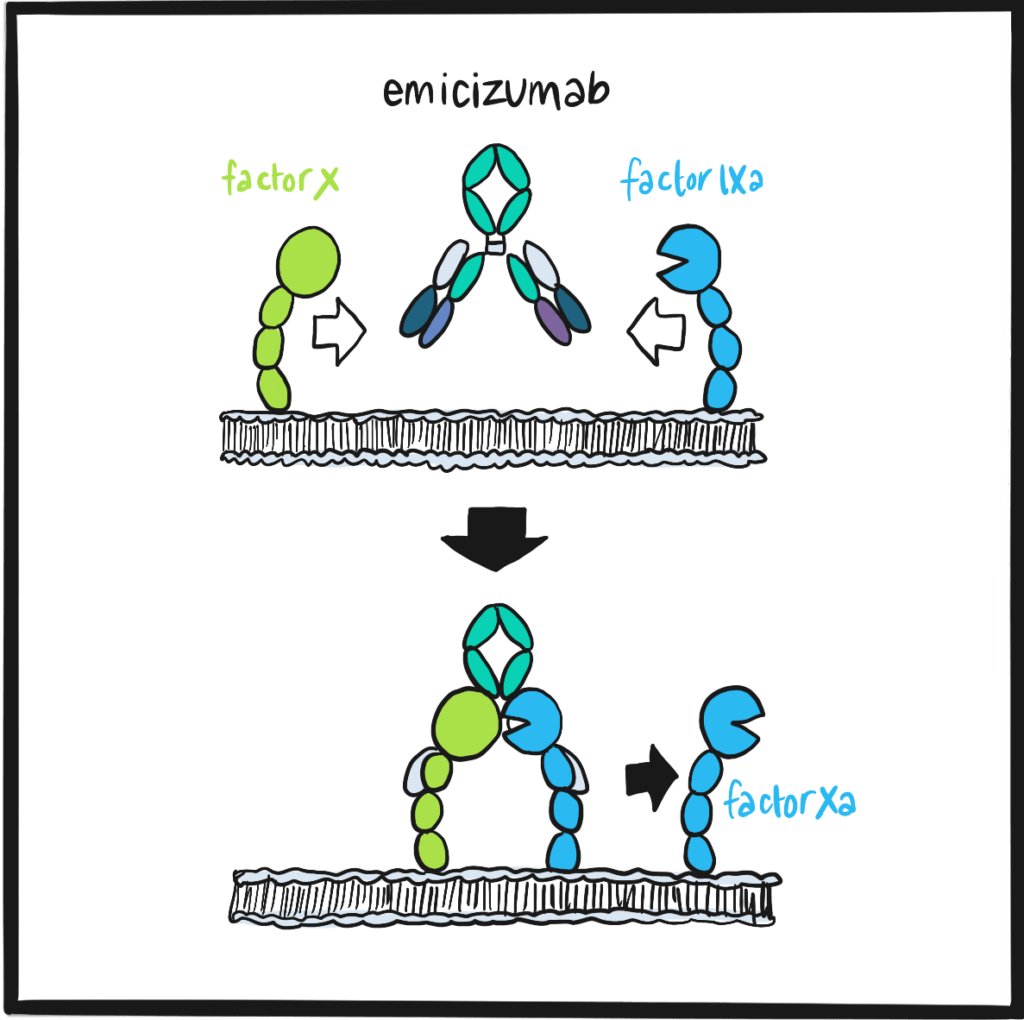
More questions than answers
Yet more switching, more paperwork, collecting QoL and HJHS scores (which means more work for nurses and physios), more bloods, more adverse event monitoring and reporting. And, of course more patient education – how to inject, which vial size, which volume, what if they bleed, making sure they have FVIII at home and that they can self infuse still – what does that mean for newly diagnosed babies whose parents can’t do this (maybe they have only had ports) or boys who will never learn to self-infuse?
And what about those with moderate haemophilia who are on prophylaxis – NHSE agreement is for severe haemophilia only – can we bend the rules or are they exempt? And how unfair is that if they are severely affected?
Oh, and this is NHSE remember – so poor Scottish, Northern Irish and Welsh patients just carry on with factor – for how long? And what if they want to move to England for new treatments – how are we going to handle that?
Don’t get me wrong – this is fantastic news but there are many ifs and buts yet to be answered. UKHCDO has offered some potentially useful tips but many of these also raise some further questions.
Before switching |
|
| UKHCDO advises that each patient has been fully informed of the need to use up the majority of their factor VIII stock but to keep a small supply in case of breakthrough bleed (NHSE request that this should usually be the best value recombinant FVIII). | So does that mean a factor switch as well – for those on plasma derived it will – what about others? |
| UKHCDO advises that usage of Haemtrack is compulsory for emicizumab doses and for all additional FVIII required. | No surprise there! |
| UKHCDO advises that usage of Haemtrack is compulsory for emicizumab doses and for all additional FVIII required. | Is this the best value FVIII? – what if the patient was on a more expensive product before switching? |
Loading phase; week 1-4 |
|
UKHCDO advises that on the day of starting the drug the patient should have baseline bloods taken and factor VIII prophylaxis should stop. The suggested schedule for hospital/home dosing through loading phase:
|
This is because the maintenance dose will be different and may come from different vials to save wastage – managing that will be interesting – how will you remember not just the dose but also the vials and volume for every patient? |
Outcome monitoring, safety and efficacy surveillance |
|
UKHCDO advises that adverse events should be reported to the NHD. The quarterly returns sheet for patients on emicizumab will be modified to include “yes/no tickboxes” to report “yes/no bleeds” and “yes/no reactions”. All patients on emicizumab have outcome assessments, these will be;
Ideally all these assessments should be available or performed prior to starting and then repeated after 6 months, then every 12 months for 3 years. |
So what about patients in centres without a physio – who will do their HJHS? And I’m guessing the EQ5D will be given out by nurses – just another exercise for you? Why not use the EQ5D-Y for the under 12s – why do we never consider their views in haemophilia research? And don’t these outcomes matter in those not having emicizumab – where is the comparative data to show improvement? |
Good luck, I’m sure this week will be a busy one with lots of questions. With time we can sort those and promote better care for (some of) those with (some kinds of) haemophilia!